Volume: 3 Issue: 1
Inhibin Hormone and Female Infertility - A Review
Year: 2026, Page: 1-7, Doi: https://doi.org/10.71325/ajjms.v3i1.2 5.89
Received: Feb. 2, 2026 Accepted: March 23, 2026 Published: March 31, 2026
Abstract
The male or female genital hormone inhibin selectively decreases the synthesis of FSH from the pituitary gland. Inhibin is often promoted as a fertility-regulating drug, especially for males, due to its ability to specifically inhibit FSH release, thereby reducing germ cell production. After summarizing current research on inhibin extraction and cDNA sequencing of inhibin mRNA, this review examines the potential of using inhibitors as a reproductive modulator and a diagnostic tool for infertility in both men and women. Isolation and identification of inhibin is challenging, and there is not much understanding of its physiological characteristics. Purification of inhibin from ovarian follicular fluid relies on selecting specific molecular weight forms of proteins. These forms, which exhibit biological inhibin activity, are identified following SDS-PAGE separation of follicular fluid proteins. Gel electrophoresis resolves protein species (e.g., 32–120 kDa forms), enabling size-based isolation and FSH-suppressive bioactivity confirmation, per foundational studies. To investigate inhibin as a prospective fertility-regulating agent, it must induce sustained and irreversible effects. If shown, this would solve key challenges for long-term polypeptide contraceptives, including administration methods, delivery management, and the risk of antibody formation, especially after prolonged use.
Keywords: Inhibin, Female Infertility, Folliculogenesis
INTRODUCTION
Inhibin has been considered as a suppressor of follicle-stimulating hormone (FSH) secretion from the anterior pituitary through pituitary-gonad negative feedback to regulate follicle development. It was demonstrated that the addition of inhibin A could significantly suppress FSH-induced FSHR mRNA level in cultured rat granulosa cells (GCs) measured by real-time polymerase chain reaction (PCR). The inhibin A exerted its action mainly by inhibiting FSHR promoter activity. Furthermore, exogenous inhibin A could dramatically decrease FSH-induced P450arom and P450scc levels and suppress progesterone and estradiol production in the cultured GCs, but it did not decrease forskolin-induced steroidogenesis, indicating that the inhibitory effect of inhibin A on FSH action may be upstream of cAMP signaling[1, 2]. The hypothalamic-pituitary-gonadal axis, which regulates reproductive steroids, requires steroidogenic factor-1 (SF-1) for its development and function. SF-1 is repressed by DAX-1, which may prevent it from activating downstream genes such as the inhibin α-subunit genes. When DAX-1 binds to SF-1, it may affect how it interacts with other co-activators as well as transcription elements[3, 4]. Androgens, acting through the androgen receptor (AR), can also influence inhibin production. In some cases, the AR can interact with SF-1 to regulate gene expression related to reproduction[5, 6]. Infertility disorders could be evaluated by quantifying the circulating levels of inhibin B. The major sites of inhibin B production are from male testicles as well as female follicles. Follicles are the tissues where viable eggs are created[7, 8]. Inhibin B levels vary across the menstrual cycle, peaking in the early follicular phase. Fertile men show higher levels than those with Sertoli cell dysfunction[9].
Structure and function of inhibin
Inhibin is a dimeric glycoprotein hormone secreted by Sertoli cells in men and granulosa cells in women, acting primarily as a negative feedback regulator. It inhibits the synthesis and release of FSH from the anterior pituitary. It consists of an α–subunit disulphide–linked to either a βA subunit (inhibin A) or βB subunit (inhibin B) and is mainly expressed by the gonads, ovarian granulosa cells in females, and testicular Sertoli cells in males[10, 11].
Quantifying serum Inhibin levels will help in assessing the pathophysiology of puberty, ovulation, menopause, and infertility, and serve as markers for ovarian sex cord-stromal tumors (e.g., granulosa cell tumors)[12].
Physiological role and mechanism of action of inhibin hormone
The ovaries generate reproductive hormones called inhibins, which are well recognized for their ability to inhibit the synthesis and secretion of FSH from the anterior pituitary gland. The primary circulatory form of inhibin in adolescent and adult males across many mammals is inhibin B[11]. The gonads are the primary sources of circulating inhibins under normal physiological conditions, as ovariectomy or castration markedly reduces or eliminates detectable levels. In vitro, inhibin B is more potent than inhibin A at suppressing FSH release (approximately 4-fold in pituitary cell models)[10, 11, 13]. When it comes to inhibiting FSH in vitro conditions, inhibin B is more effective than inhibin A. During menstrual cycles, levels of both inhibin A and inhibin B are regulated in different ways. Across the luteal-follicular transition, inhibin B rises (peaking early in the follicular phase around cycle day 7), while inhibin A remains low until the late follicular phase and dominates the luteal phase[14, 15]. When a woman is not pregnant, the corpus luteum deteriorates with a fall in inhibin A levels. The next one starts when these strong adverse reaction signals are absent, causing systemic FSH levels to rise[16].
In rats, inhibin B is elevated on metestrus/diestrus cycles (with low FSH), then declines late proestrus alongside inhibin A (which rises from low metestrus to proestrus peak). Both drops in the estrus cycle before the FSH surge recruit new follicles[17, 18]. Both inhibins are produced by ovarian granulosa cells in follicles across species. Low inhibin A or B during the estrus cycle reflects an early rise in FSH in women, aiding follicle development for potential ovulation[18, 19].
Role of Inhibin in Female Fertility
Inhibin B is a key hormone for fertility in both males and females. Serum levels of inhibin B are measured via immunoassays such as the Enzyme-Linked immunosorbent assay (ELISA), typically on days two to three of the menstrual cycle in women to assess ovarian reserve or infertility, and at any time in men to evaluate spermatogenesis[19, 20]. Two AMH patterns align with inhibin B trends: “aging ovary” pattern includes a low/stable AMH, low inhibin B, short 18-21 day cycles, and reduced follicle recruitment. While “younger ovary” with a higher AMH with days 2-7 peaks, where >1 ng/mL requires cycle-day adjustment for accurate interpretation alongside inhibin B[21].
-
Inhibin B is linked to the development of the ovaries within females and suppresses FSH during the early follicular phase.
-
In men, inhibin B from Sertoli cells supports spermatogenesis. Fertile males with normal conception rates typically show higher serum inhibin B than those with oligospermia or impaired sperm production.
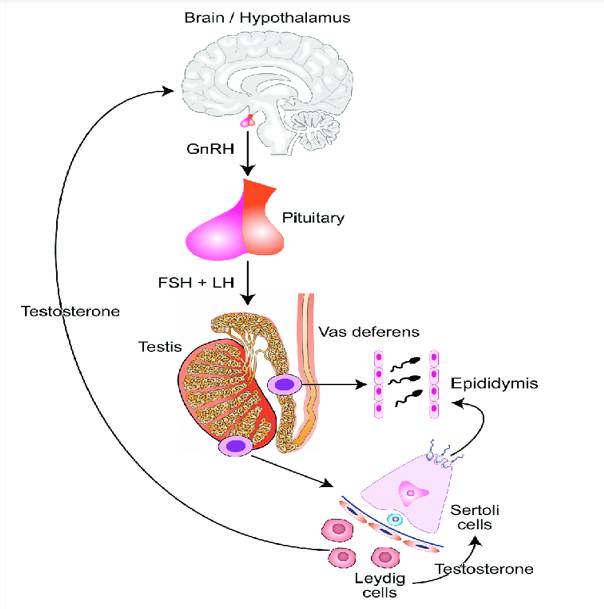
Fig. 1: The hypothalamic-pituitary-testicular axis and its role in spermatogenesis. CCBY 4.0 License -John Reynolds-Wright. Image accessible via https://flic.kr/p/2hgvk5V6A. FSH, follicle-stimulating hormone; GnRH, gonadotropin-releasing hormone; LH, luteinizing hormone[22]
[Fig. 1] shows the regulation of gonadotropin release by activins and inhibins. Inhibins exert their inhibitory effect by selectively suppressing the secretion of FSH from the anterior pituitary gland[23]. FSH’s significant role in reproductive function and the ability of inhibin to steadily reduce FSH levels below the threshold that is essential for gametogenesis. Hence, inhibin has potential utility as a fertility-regulating agent[24]. No studies have investigated this mechanism in vivo across species due to the past unavailability of pure, authentic inhibin preparations for such a study[22].
Diagnostic and therapeutic applications
Inhibins are part of the Transforming growth factor -β (TGF-β) superfamily, which includes hormones that have multiple roles in development and reproduction. Inhibins, unlike other members of the family, have a restricted tissue distribution, are principally expressed in the gonadal and placental tissues, and have tissue-specific effects. Inhibins, which are best known for their negative feedback control of FSH release from anterior pituitary gonadotrophs, have lately been linked to other functions, including tumor suppression in gonadal and reproductive organs[25, 26].
Maternal serum inhibin A levels are elevated in Down syndrome (trisomy 21) pregnancies and serve as an important second-trimester biomarker (typically weeks 15-20), improving detection rates when combined with alpha-fetoprotein, hCG, and estriol in quadruple tests (e.g., detection sensitivity ~75-80% with 5% false-positive rate)[27].
The quadruple test, which involves screening maternal blood for inhibin A, α-fetoprotein, unconjugated estriol, and human chorionic gonadotropin (hCG), detects roughly 70-75% of pregnancies with Down syndrome, whereas the double test (α-fetoprotein and hCG alone) only detects about 50-60%[28, 29]. Although varied cutoffs restrict their normal clinical usage, low inhibin B levels in early pregnancy, especially in assisted reproductive technologies, predict poor ovarian response[30]. As a miscarriage biomarker, inhibin A has been evaluated; it is good at detecting very early losses, but it is not an accurate indicator of miscarriages that occur after about six weeks[31]. Total inhibin levels are used to detect ex cord-stromal tumors (5–10% of ovarian neoplasms) with nearly 100% sensitivity; among epithelial ovarian malignancies, 5–35% of non-mucinous subtypes and up to 80% of mucinous instances exhibit elevations, enabling continuous tumor subtyping investigation[32, 33].
Patients seeking assisted reproductive technologies (ART) receive a speedy, exact evaluation of their chances of becoming pregnant and beginning specialized therapies[34]. Ovarian reserve testing is an important part of the female infertility workup before ART[34, 35]. Success in techniques such as intracytoplasmic sperm injection (ICSI) and in vitro fertilization (IVF) is dependent on the ovaries' capacity to respond to hormonal stimulation by recruiting numerous follicles[35]. Female subfertility is frequently caused by hyper- or hypogonadotropic dysovulation, such as the most common type, polycystic ovary syndrome (PCOS), or age-related ovarian malfunction[36, 37].
Diminished Ovarian Reserve
The number of oocytes remaining in the ovaries, which reflects the reproductive potential of a woman, is termed ovarian reserve. Numerous factors, including hormones, metabolites, baseline ovarian reserve, disease conditions, environmental factors, and medications, may influence ovarian reserve[38]. Decreased ovarian reserve (DOR) has emerged as one of the most prevalent issues in contemporary therapeutic reproductive medicine due to the tendency of delaying conception throughout contemporary culture. Because of its complicated clinical characteristics as well as uncertain process, doctors find it challenging to provide tailored therapy[34]. This review addresses the factors affecting ovarian reserve as well as explores the pathogenic variables or attainable impacts that might explain the possible pathways of DOR.
Polycystic Ovary Syndrome
Despite the paucity of evidence on the function of inhibin in pathophysiologic conditions, several interesting findings have been reported. In polycystic ovarian syndrome (PCOS), basal and early/mid-follicular inhibin levels are characteristically similar to those of controls. Still, they fail to rise in the late follicular, midcycle, or luteal phases due to anovulation and the lack of a dominant follicle[39].
Exogenous gonadotropin stimulation causes a normal inhibin response in PCOS ovaries, according to studies. These findings indicated intact ovarian physiology and that gonadotropin dysregulation, rather than intrinsic abnormalities, is the cause of changes in inhibin levels[40, 41]. It has been shown that a luteal phase confirmed by biopsy was linked to extremely low serum inhibin concentrations in both the follicular and luteal phases[42].
According to studies, higher inhibin levels were associated with increased androgens in PCOS, particularly inhibin B, which may improve LH-mediated androgen synthesis via granulosa-theca interactions[43, 44].
Overall, the circulating inhibin levels show ovarian follicular and luteal activity throughout the female life span. They increase with puberty, indicating activation of granulosa cells. During reproductive years, they show cyclical patterns (inhibin B peaking in the follicular phase from small antral follicles, inhibin A in the luteal phase from the corpus luteum). The levels decrease with ovarian aging because of a declining follicle reserve. A higher level of serum inhibin, especially inhibin A or B, may be a sign of granulosa cell tumors or subtypes of PCOS, among other ovarian diseases. Gonadally generated inhibin promotes Folliculogenesis and steroidogenesis in the ovary through autocrine and paracrine actions, while also exerting systemic negative feedback on pituitary FSH output[45-47].
Primary Ovarian Insufficiency
The depletion or dysfunction of ovarian follicles, resulting in oligo- or amenorrhea before age 40 years, is generally considered as primary ovarian insufficiency (POI). No universal diagnostic criteria are available for POI in adolescents, leading to delays in diagnosis. Post diagnosis, it is recommended annually to monitor bone density, cardiovascular risk, and psychological health. Hormone therapy might help to reduce vasomotor or urogenital symptoms and ease long-term risks to bone health, cardiovascular disease, and sexual function[48, 49]. The ovarian morphology and histology of POI are distinct from those of the normal ovary [Fig. 2].
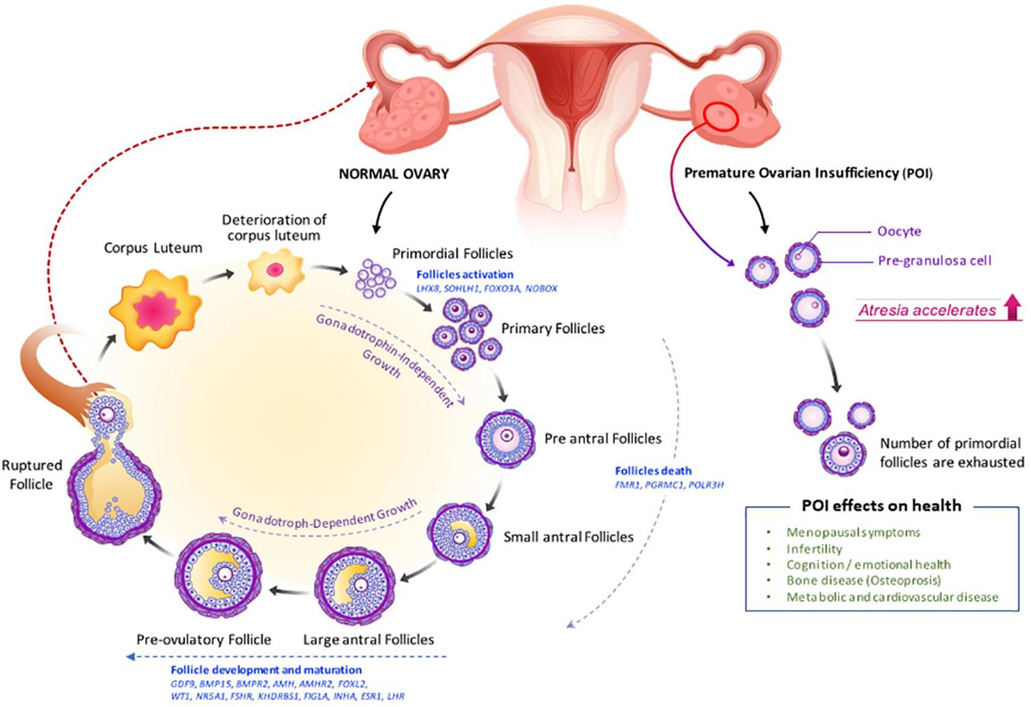
Fig. 2: Folliculogenesis and ovulation in a normal ovary versus POI[48]
For fertility preservation or reproductive treatments, it is advised to refer the patients or families to an endocrinologist as well as an infertility expert if required. Primary ovarian insufficiency (POI) affects approximately 1 in 1,000 women by age 30 and 1 in 100 by age 40, showing as oligo/amenorrhea, infertility, hypoestrogenism, and elevated gonadotropins before age 40. Women with POI often present with high blood FSH (>25 IU/L on two occasions), low estradiol (<50 pg/mL), and decreased inhibin B (indicating depleted antral follicles); however, inhibin A may vary[50, 51].
Endometriosis and Ovarian Dysfunction
Endometriosis affects about 10-15 % of women of reproductive age. Among women with endometriosis, 30 to 50% are infertile[52]. However, the exact etiology of infertility linked to endometriosis is still unknown. Endometriosis-related infertility may be due to difficulty getting pregnant or maintaining a healthy pregnancy. Although the precise mechanisms causing early or premature menopause in endometriosis-affected women are still not fully understood, growing evidence points to a link between endometriosis and both an earlier menopausal start and a decreased ovarian reserve[53, 54]. Endocrinology-linked infertility is heterogeneous. Morphological abnormalities triggered by adherent or pelvic masses can be the root cause of sterility. In contrast to the structural impediments, abnormal ovarian functioning in women with endometriosis might contribute to reduced fertility. Epidemiologic studies indicate that women with endometriosis, particularly those with ovarian endometriomas and associated surgery, are more likely to experience a faster reduction in ovarian viability and an earlier natural menopause, even though the causality is not fully understood[53, 54].
Three fundamental components comprise ovarian function:
(1) Ovarian hormone production,
(2) Preservation of follicular growth from the preantral to the luteal stage after ovulation, or
(3) Inactive oocyte reserve[54]
Clinical Applications of Inhibin in Reproductive Medicine
The evaluation of female ovarian reserve or biological capability is two of the many therapeutic applications for inhibin B. It could also help track the beginning of the menopausal transition, gender-related abnormalities, and ovarian recovery after chemotherapy[55].
The cerebellum, genitals, and the glandular system are just a few of the numerous components involved in the intricate processes that control the human reproductive mechanism. The granulosa of the ovary and the Sertoli cells of the testis release this polypeptide hormone. Semen levels or testicular volume are favorably correlated with plasma inhibin B levels. Inhibin B could be significant as a measure of Sertoli cell activity in infertile men as well as a prognosis predictor among women receiving ovulation-promoting medication, according to present knowledge of inhibin metabolism and pathophysiology in humans. The inhibin α-subunit was elevated in aggressive prostate diseases[56]. The nature, oversight, and therapeutic applications of inhibin and several similar compounds are covered in this article.
Potential applications have been evaluated in 4 distinct fields: ovarian cancer, PCOS, the ovarian reserve, including operation, or childbirth, and pre-eclampsia with Down syndrome screening. Inhibin physiology, as its therapeutic significance in reproductive healthcare, has been the subject of investigation in recent years. Inhibin A and B are synthesized from the gonads during gametogenesis and exhibit distinct release patterns in women across the reproductive cycle. Additionally, inhibins contribute to pregnancy's biological adaptation since they are also produced by the placental and embryonic tissues. The most recent developments about the role of inhibins in human reproduction are covered in this review. Inhibin A and B have led to significant advancements in our knowledge of reproductive inhibitors[57]. The review discussed the therapeutic application of measuring inhibin levels throughout the blood and various physiological fluids. Studies have identified changes in inhibin concentrations in gynecological disorders as well as in normal or pathogenic pregnancies. In women, the tissue of the corpus luteum or the dominant ovarian follicles is the main producer of inhibin A, while tiny emerging follicles are the main producers of inhibin B. This menstrual cycle causes fluctuations in plasma inhibin A or B levels. Serum concentrations of inhibin A and B drop to extremely low or undetected amounts after menopause due to the depletion of ovarian follicles. Current research has shown that inhibin A may be a more accurate indicator of maternal functioning than human chorionic gonadotropin (hCG) due to its short half-life, even though its exact physiological role during childbirth is unknown. If inhibin A levels are measured earlier in pregnancy, they may be used to anticipate premature delivery, Down syndrome, and fetal development constraints throughout the first and/or second trimester before clinical manifestations appear.
CONCLUSION
The glycoprotein hormone inhibin exerts negative feedback on FSH secretion from the anterior pituitary gland. Their secretion from granulosa cells inversely correlated to FSH levels throughout the estrogen cycle. Immunization against inhibin raises the production of FSH as well as the growth of follicles, which in turn raises the fertilization rates. It has been demonstrated that inhibins can be in two forms by dimeric construction of an α-subunit, as well as one of two extremely associated β-subunits to create inhibin A (α- βA), as well as inhibin B (α- βB). While inhibin components are expressed in numerous organs, reproductive organs are the principal source of circulating inhibins. Though they can act as a paracrine or autocrine component in certain tissues, their best-recognized activities are as endocrine controllers of hypothalamic GnRH.
This review focused on more recent advancements in inhibin research, characterizing variations in inhibin A and B emission throughout the menstrual cycle. We also examine vaccination against inhibin α subunit as a viable technique for superovulation. Super ovulation has been produced effectively by passive or active vaccination against the inhibin α-subunit in numerous mammals. Furthermore, multiple studies have indicated that embryos super ovulated with vaccination to inhibin α-subunit possess the potential to mature properly, indicating that inhibin-based immunization might be employed as a feasible strategy for superovulation within a wide variety of vertebrate populations.
DISCLOSURE
Conflict of interest: The authors declare no conflicts of interest.
Funding: Nil.
References
1. Cook RW, Thompson TB, Jardetzky TS, Woodruff TK. Molecular Biology of Inhibin Action. Seminars in Reproductive Medicine. 2004; 22 (3). Available from: https://doi.org/10.1055/s-2004-831902
2. Suntharalingham JP, Buonocore F, Duncan AJ, Achermann JC. DAX-1 (NR0B1) and steroidogenic factor-1 (SF-1, NR5A1) in human disease. Best Practice & Research Clinical Endocrinology & Metabolism. 2015; 29 (4). Available from: https://doi.org/10.1016/j.beem.2015.07.004
3. Mukai T, Kusaka M, Kawabe K, Goto K, Nawata H, Fujieda K, <I>et al</I>. Sexually dimorphic expression of Dax‐1 in the adrenal cortex. Genes to Cells. 2002; 7 (7). Available from: https://doi.org/10.1046/j.1365-2443.2002.00556.x
4. Elzenaty NR, Kouri C, de Lapiscina MI, Sauter K S, Moreno F, Tarruella CN, <I>et al</I>. NR5A1/SF-1 Collaborates with Inhibin α and the Androgen Receptor. International Journal of Molecular Sciences. 2024; 25 (18). Available from: https://doi.org/10.3390/ijms251810109
5. Edelsztein NY, Rey RA. Importance of the Androgen Receptor Signaling in Gene Transactivation and Transrepression for Pubertal Maturation of the Testis. Cells. 2019; 8 (8). Available from: https://doi.org/10.3390/cells8080861
6. Gregory SJ, Kaiser UB. Regulation of Gonadotropins by Inhibin and Activin. Seminars in Reproductive Medicine. 2004; 22 (3). Available from: https://doi.org/10.1055/s-2004-831901
7. Kumanov P, Nandipati K, Tomova A, Agarwal A. Inhibin B is a better marker of spermatogenesis than other hormones in the evaluation of male factor infertility. Fertility and Sterility. 2006; 86 (2). Available from: https://doi.org/10.1016/j.fertnstert.2006.01.022
8. Lockwood G. The Diagnostic Value of Inhibin in Infertility Evaluation. Seminars in Reproductive Medicine. 2004; 22 (3). Available from: https://doi.org/10.1055/s-2004-831895
9. Kong X, Ye Z, Chen Y, Zhao H, Tu J, Meng T, <I>et al</I>. Clinical application value of Inhibin B alone or in combination with other hormone indicators in subfertile men with different spermatogenesis status: A study of 324 Chinese men. Journal of Clinical Laboratory Analysis. 2021; 35 (8). Available from: https://doi.org/10.1002/jcla.23882
10. Explore Anatomy. <I>Inhibin - Structure, Function & Location</I> [Internet]. [place unknown]: Explore Anatomy; c2025 [cited 2026 Feb 13]. Available from: https://exploreanatomy.com/endocrine-system/inhibin
11. Walton K L, Goney M P, Peppas Z, Stringer J M, Winship A, Hutt K, <I>et al</I>. Inhibin Inactivation in Female Mice Leads to Elevated FSH Levels, Ovarian Overstimulation, and Pregnancy Loss. Endocrinology. 2022; 163 (4). Available from: https://doi.org/10.1210/endocr/bqac025
12. Makanji Y, Zhu J, Mishra R, Holmquist C, Wong W P, Schwartz N B, <I>et al</I>. Inhibin at 90: From Discovery to Clinical Application, a Historical Review. Endocrine Reviews. 2014; 35 (5). Available from: https://doi.org/10.1210/er.2014-1003
13. Cobellis L, Luisi S, Pezzani I, Reis FM, De Leo V, Petraglia F. Serum inhibin A, inhibin B, and pro-αC levels are altered after surgically or pharmacologically induced menopause. Fertility and Sterility. 2002; 77 (4). Available from: https://doi.org/10.1016/s0015-0282(01)03234-4
14. Welt CK, Smith ZA, Pauler DK, Hall JE. Differential Regulation of Inhibin A and Inhibin B by Luteinizing Hormone, Follicle-Stimulating Hormone, and Stage of Follicle Development<sup>1</sup>. The Journal of Clinical Endocrinology & Metabolism. 2001; 86 (6). Available from: https://doi.org/10.1210/jcem.86.6.7597
15. Reed BG, Carr BR. TThe normal menstrual cycle and the control of ovulation. In: Feingold KR, Anawalt B, Blackman MR, et al., editors. <I>Endotext</I>. South Dartmouth (MA): MDText.com, Inc.; 2000. Available from: https://www.ncbi.nlm.nih.gov/books/NBK279054/
16. Thakur MP, Reddy KS. Inhibin and activin in ovarian physiology. Journal of Endocrinology. 2018; 237(2): R47–R61.
17. Woodruff TK, Besecke LM, Groome N, Draper LB, Schwartz NB, Weiss J. Inhibin A and inhibin B are inversely correlated to follicle-stimulating hormone, yet are discordant during the follicular phase of the rat estrous cycle, and inhibin A is expressed in a sexually dimorphic manner.. Endocrinology. 1996; 137 (12). Available from: https://doi.org/10.1210/endo.137.12.8940372
18. Walton KL, Goney MP, Peppas Z, Stringer JM, Winship A, Hutt K, <I>et al</I>. Inhibin Inactivation in Female Mice Leads to Elevated FSH Levels, Ovarian Overstimulation, and Pregnancy Loss. Endocrinology. 2022; 163 (4). Available from: https://doi.org/10.1210/endocr/bqac025
19. Yamoto M, Imai M, Otani H, Nakano R. Serum Levels of Inhibin A and Inhibin B in Women With Normal and Abnormal Luteal Function. Obstetrics & Gynecology. 1997; 89 (5). Available from: https://doi.org/10.1016/s0029-7844(97)00081-1
20. Gutiérrez A, Muñoz-Pérez R, Zapater P, Mira C, Rodríguez A, Sempere-Robles L, <I>et al</I>. Inhibin B and antiMüllerian hormone as surrogate markers of fertility in male and female Crohn’s disease patients: a case-control study. Frontiers in Medicine. 2024; 11 Available from: https://doi.org/10.3389/fmed.2024.1374603
21. Jankowska K, Suszczewicz N, Rabijewski M, Dudek P, Zgliczyński W, Maksym RB. Inhibin-B and FSH Are Good Indicators of Spermatogenesis but Not the Best Indicators of Fertility. Life. 2022; 12 (4). Available from: https://doi.org/10.3390/life12040511
22. Reynolds-Wright JJ, Anderson RA. Male contraception: where are we going and where have we been?. BMJ Sexual & Reproductive Health. 2019; 45 (4). Available from: https://doi.org/10.1136/bmjsrh-2019-200395
23. Lu C, Yang W, Chen M, Liu T, Yang J, Tan P. Inhibin A inhibits follicle-stimulating hormone (FSH) action by suppressing its receptor expression in cultured rat granulosa cells. Molecular and Cellular Endocrinology. 2009; 298 (1-2). Available from: https://doi.org/10.1016/j.mce.2008.09.039
24. Lin YF, Brûlé E, Ongaro L, Zhou X, Jin Y, Schultz H. Loss of Inhibin Negative Feedback to Pituitary Gonadotropes Leads to Enhanced Ovulation but Pregnancy Failure in Mice. Endocrinology. 2025; 166 (10). Available from: https://doi.org/10.1210/endocr/bqaf142
25. Namwanje M, Brown CW. Activins and Inhibins: Roles in Development, Physiology, and Disease. Cold Spring Harbor Perspectives in Biology. 2016; 8 (7). Available from: https://doi.org/10.1101/cshperspect.a021881
26. Knight PG, Glister C. TGF-β superfamily members and ovarian follicle development. Reproduction. 2006; 132 (2). Available from: https://doi.org/10.1530/rep.1.01074
27. Christiansen M, Nørgaard-Pedersen B. Inhibin A is a maternal serum marker for Down's syndrome early in the first trimester. Clinical Genetics. 2005; 68 (1). Available from: https://doi.org/10.1111/j.1399-0004.2005.00441.x
28. Harrison G, Goldie D. Second-trimester Down's syndrome serum screening: double, triple or quadruple marker testing?. Annals of Clinical Biochemistry: International Journal of Laboratory Medicine. 2006; 43 (1). Available from: https://doi.org/10.1258/000456306775141876
29. Li Y, Zhang X, Sun Y, Hong D, Wang Y, Xu Z, <I>et al</I>. Combined detection of α-fetoprotein and free β-human chorionic gonadotropin in screening for trisomy 21 and management of cases in the moderate risk value range. Molecular and Clinical Oncology. 2017; 7 (4). Available from: https://doi.org/10.3892/mco.2017.1355
30. Fawzy M, Lambert A, Harrison R F, Knight P G, Groome N, Hennelly B, <I>et al</I>. Day 5 inhibin B levels in a treatment cycle are predictive of IVF outcome. Human Reproduction. 2002; 17 (6). Available from: https://doi.org/10.1093/humrep/17.6.1535
31. Muttukrishna S, Jauniaux E, Greenwold N, McGarrigle H, Jivraj S, Carter S, <I>et al</I>. Circulating levels of inhibin A, activin A and follistatin in missed and recurrent miscarriages. Human Reproduction. 2002; 17 (12). Available from: https://doi.org/10.1093/humrep/17.12.3072
32. Robertson D M, Pruysers E, Jobling T. Inhibin as a diagnostic marker for ovarian cancer. Cancer Letters. 2007; 249 (1). Available from: https://doi.org/10.1016/j.canlet.2006.12.017
33. Tsigkou A, Marrelli D, Reis FM, Luisi S, Silva-Filho AL. Total Inhibin Is a Potential Serum Marker for Epithelial Ovarian Cancer. The Journal of Clinical Endocrinology & Metabolism. 2007; 92 (7). Available from: https://doi.org/10.1210/jc.2007-0235
34. Mutlu MF, Erdem A. Evaluation of ovarian reserve in infertile patients. Journal of the Turkish German Gynecological Association. 2012; 13 (3). Available from: https://doi.org/10.5152/jtgga.2012.28
35. Practice Committee of the American Society for Reproductive Medicine. Testing and interpreting measures of ovarian reserve: a committee opinion. Fertility and Sterility. 2012; 98 (6). Available from: https://doi.org/10.1016/j.fertnstert.2012.09.036
36. Cecchino GN, Canillas GM, Cruz M, García-Velasco JA. Impact of hypogonadotropic hypogonadism on ovarian reserve and response. Journal of Assisted Reproduction and Genetics. 2019; 36 (11). Available from: https://doi.org/10.1007/s10815-019-01587-7
37. Cunha A, Póvoa AM. Infertility management in women with polycystic ovary syndrome: a review. Porto Biomedical Journal. 2021; 6 (1). Available from: https://doi.org/10.1097/j.pbj.0000000000000116
38. Ulrich ND, Marsh EE. Ovarian Reserve Testing: A Review of the Options, Their Applications, and Their Limitations. Clinical Obstetrics & Gynecology. 2019; 62 (2). Available from: https://doi.org/10.1097/grf.0000000000000445
39. Buckler HM, McLachlan RI, MacLachlan VB, Healy DL, Burger HG. Serum Inhibin Levels in Polycystic Ovary Syndrome: Basal Levels and Response to Luteinizing Hormone-Releasing Hormone Agonist and Exogenous Gonadotropin Administration*. The Journal of Clinical Endocrinology & Metabolism. 1988; 66 (4). Available from: https://doi.org/10.1210/jcem-66-4-798
40. Dafopoulos K, Venetis C, Messini CI, Pournaras S, Anifandis G, Garas A, <I>et al</I>. Inhibin secretion in women with the polycystic ovary syndrome before and after treatment with progesterone. Reproductive Biology and Endocrinology. 2011; 9 (1). Available from: https://doi.org/10.1186/1477-7827-9-59
41. Wachs DS, Coffler MS, Malcom PJ, Shimasaki S, Chang RJ. Increased Androgen Response to Follicle-Stimulating Hormone Administration in Women with Polycystic Ovary Syndrome. The Journal of Clinical Endocrinology & Metabolism. 2008; 93 (5). Available from: https://doi.org/10.1210/jc.2007-2664
42. Soules MR, McLachlan RI, Marit EK, Dahl KD, Cohen NL, Bremner WJ. Luteal Phase Deficiency: Characterization of Reproductive Hormones over the Menstrual Cycle*. The Journal of Clinical Endocrinology & Metabolism. 1989; 69 (4). Available from: https://doi.org/10.1210/jcem-69-4-804
43. Shayya RF, Rosencrantz MA, Chuan SS, Cook-Andersen H, Roudebush WE, Irene Su H, <I>et al</I>. Decreased inhibin B responses following recombinant human chorionic gonadotropin administration in normal women and women with polycystic ovary syndrome. Fertility and Sterility. 2014; 101 (1). Available from: https://doi.org/10.1016/j.fertnstert.2013.09.037
44. Rambaran N, Islam MS. Decoding androgen excess in polycystic ovary syndrome: Roles of insulin resistance and other key intraovarian and systemic factors. World Journal of Diabetes. 2025; 16 (7). Available from: https://doi.org/10.4239/wjd.v16.i7.108789
45. Hauzman EE, Fancsovits P, Murber A, Rabe T, Strowitzki T, Papp Z, <I>et al</I>. Luteal-phase inhibin A and follicular-phase inhibin B levels are not characteristic of patients with an elevated LH-to-FSH ratio. Journal of Assisted Reproduction and Genetics. 2006; 23 (3). Available from: https://doi.org/10.1007/s10815-006-9043-3
46. Andersen CY. Inhibin-B secretion and FSH isoform distribution may play an integral part of follicular selection in the natural menstrual cycle. Molecular Human Reproduction. 2017; 23 (1). Available from: https://doi.org/10.1093/molehr/gaw070
47. Luisi S, Florio P, Reis FM, Petraglia F. Inhibins in female and male reproductive physiology: role in gametogenesis, conception, implantation and early pregnancy. Human Reproduction Update. 2005; 11 (2). Available from: https://doi.org/10.1093/humupd/dmh057
48. Chon SJ, Umair Z, Yoon MS. Premature Ovarian Insufficiency: Past, Present, and Future. Frontiers in Cell and Developmental Biology. 2021; 9 Available from: https://doi.org/10.3389/fcell.2021.672890
49. Sopiarz N, Sparzak PB. <I>Primary ovarian insufficiency</I>. In StatPearls. Treasure Island (FL): StatPearls Publishing LLC.2024
50. Soudeh Khanamani Falahati-Pour, Soheila Pourmasumi, Elham Sadat Mirhashemi. Ovarian Endocrine Status and ART 0utcomes in Women within PCOS Based on Different Testosterone Levels. Indian Journal of Endocrinology and Metabolism. 2023; 27 (5). Available from: https://doi.org/10.4103/ijem.ijem_360_22
51. Federici S, Rossetti R, Moleri S, Munari EV, Frixou M, Bonomi M, <I>et al</I>. Primary ovarian insufficiency: update on clinical and genetic findings. Frontiers in Endocrinology. 2024; 15 Available from: https://doi.org/10.3389/fendo.2024.1464803
52. Parasar P, Ozcan P, Terry KL. Endometriosis: Epidemiology, Diagnosis and Clinical Management. Current Obstetrics and Gynecology Reports. 2017; 6 (1). Available from: https://doi.org/10.1007/s13669-017-0187-1
53. Kulkarni MT, Shafrir A, Farland LV, Terry KL, Whitcomb BW, Eliassen AH, <I>et al</I>. Association Between Laparoscopically Confirmed Endometriosis and Risk of Early Natural Menopause. JAMA Network Open. 2022; 5 (1). Available from: https://doi.org/10.1001/jamanetworkopen.2021.44391
54. Kitajima M, Kanako M, Itsuki K, Ayumi H, Miyashita N, Matsumura A, <I>et al</I>. The Effects of Endometriosis on Ovarian Functions. Endocrines. 2021; 2 (2). Available from: https://doi.org/10.3390/endocrines2020014
55. Wen J, Huang K, Du X, Zhang H, Ding T, Zhang C, <I>et al</I>. Can Inhibin B Reflect Ovarian Reserve of Healthy Reproductive Age Women Effectively?. Frontiers in Endocrinology. 2021; 12 Available from: https://doi.org/10.3389/fendo.2021.626534
56. Balanathan P, Williams ED, Wang H, Pedersen JS, Horvath LG, Achen MG, <I>et al</I>. Elevated level of inhibin-α subunit is pro-tumourigenic and pro-metastatic and associated with extracapsular spread in advanced prostate cancer. British Journal of Cancer. 2009; 100 (11). Available from: https://doi.org/10.1038/sj.bjc.6605089
57. Medan MS, Arai KY, Watanabe G, Taya K. Inhibin: Regulation of reproductive function and practical use in females. Animal Science Journal. 2007; 78 (1). Available from: https://doi.org/10.1111/j.1740-0929.2006.00399.x
Copyright
© 2026 Published by Laxmi Memorial Education Trust. This is an open-access article under CC BY 4.0 license. (https://creativecommons.org/licenses/by/4.0/)
Cite this article
Hana Abdul-Qader Khuder, Shaymaa A H Jasim, Azzawi M Hadi, Bahaa Abdullah Laftaah ALRubaii. Inhibin Hormone and Female Infertility - A Review. AJ J Med Sci 2026;3(1):1-7